Assembly of the SIR Complex and Its Regulation by O-Acetyl-ADP-Ribose, a Product of NAD-Dependent Histone Deacetylation - ScienceDirect

The Sir4 H‐BRCT domain interacts with phospho‐proteins to sequester and repress yeast heterochromatin | The EMBO Journal

Schematic view of the structural and functional domains identified in... | Download Scientific Diagram

Assembly of the SIR Complex and Its Regulation by O-Acetyl-ADP-Ribose, a Product of NAD-Dependent Histone Deacetylation - ScienceDirect
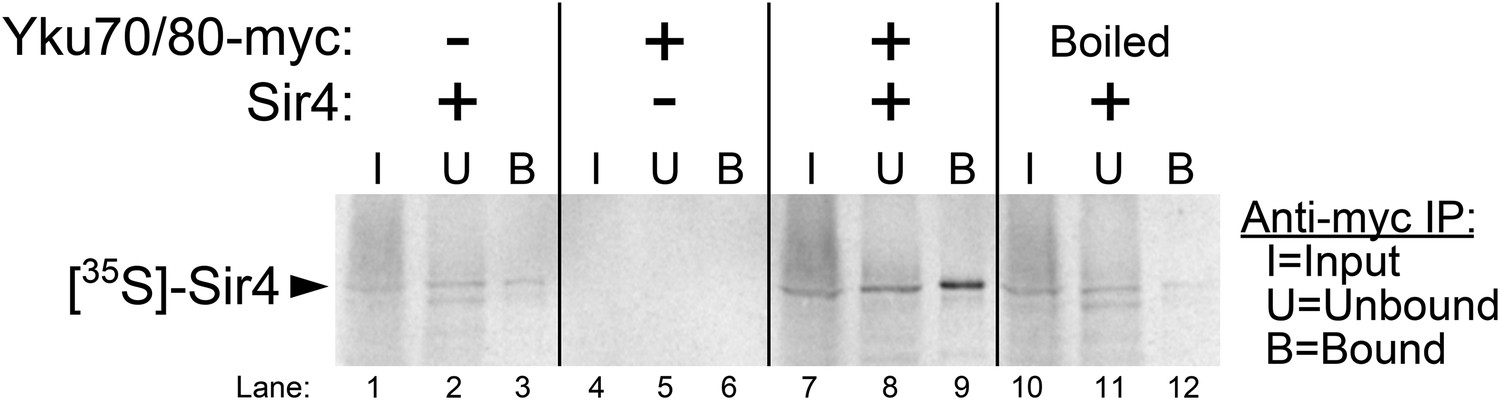
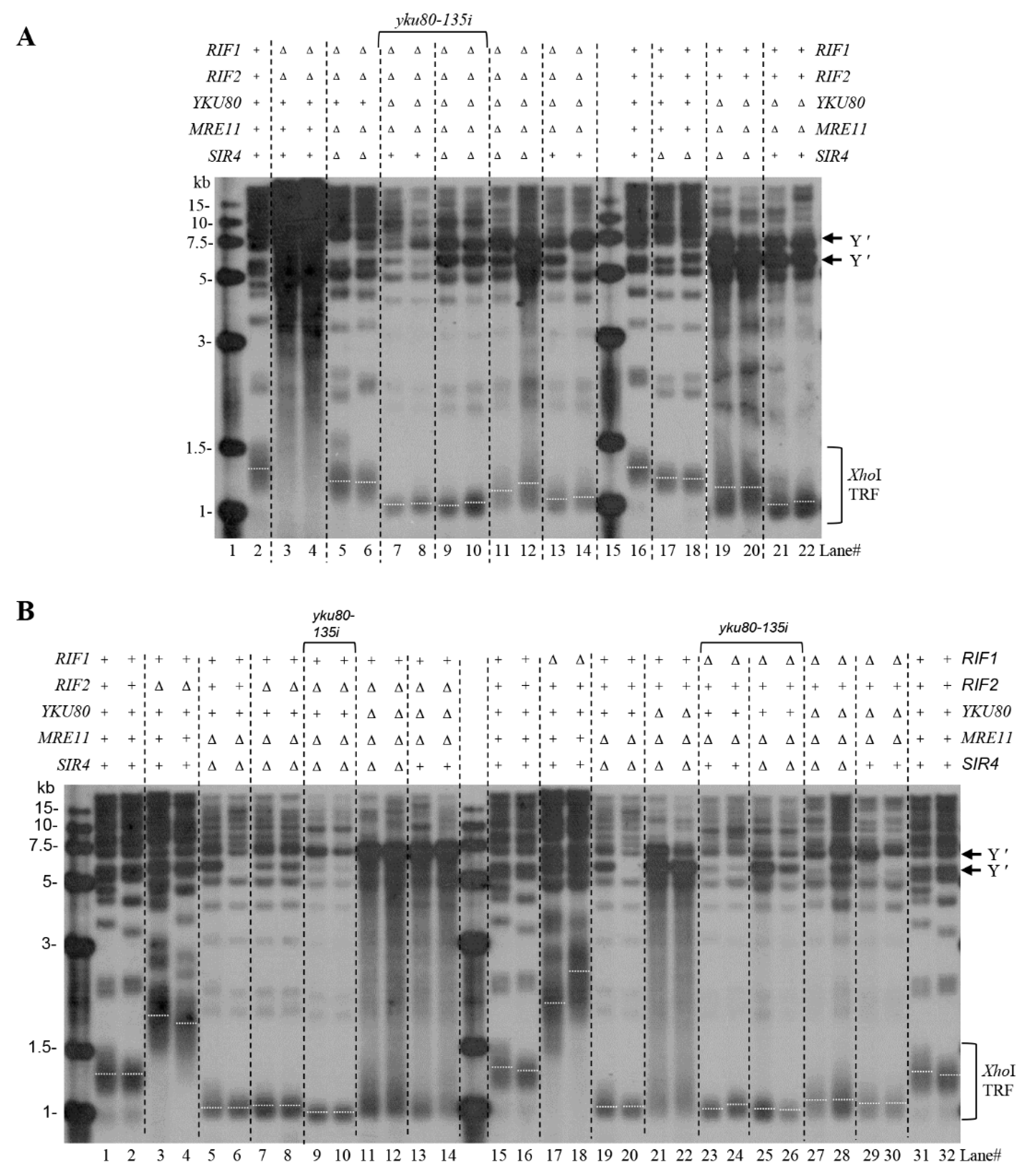
The Ku subunit of telomerase binds Sir4 to recruit telomerase to lengthen telomeres in S. cerevisiae | eLife

The Ku subunit of telomerase binds Sir4 to recruit telomerase to lengthen telomeres in S. cerevisiae | eLife

Silent information regulator protein complexes in Saccharomyces cerevisiae: a SIR2/SIR4 complex and evidence for a regulatory domain in SIR4 that inhibits its interaction with SIR3. - Abstract - Europe PMC